Projects
Antibodies! The adaptive immune response can provide perfectly fitting antibodies for anything you can imagine, given enough time, repetitive exposure and some magic. Surprisingly, there are still many black boxes in the process of antibody generation.
Core aim:
We aim to understand the regulation and mechanism of antigen-driven selection during B cell affinity maturation.
Simple is rarely easy:
High-affinity antibodies are a hallmark of effective vaccination, can provide long-lasting protection and are affinity matured in the germinal center (GC). In a bird's-eye view humoral adaptive immunity is well understood, but the little unknown details are crucial to understand the difference between failure and success. Little things make big things happen.
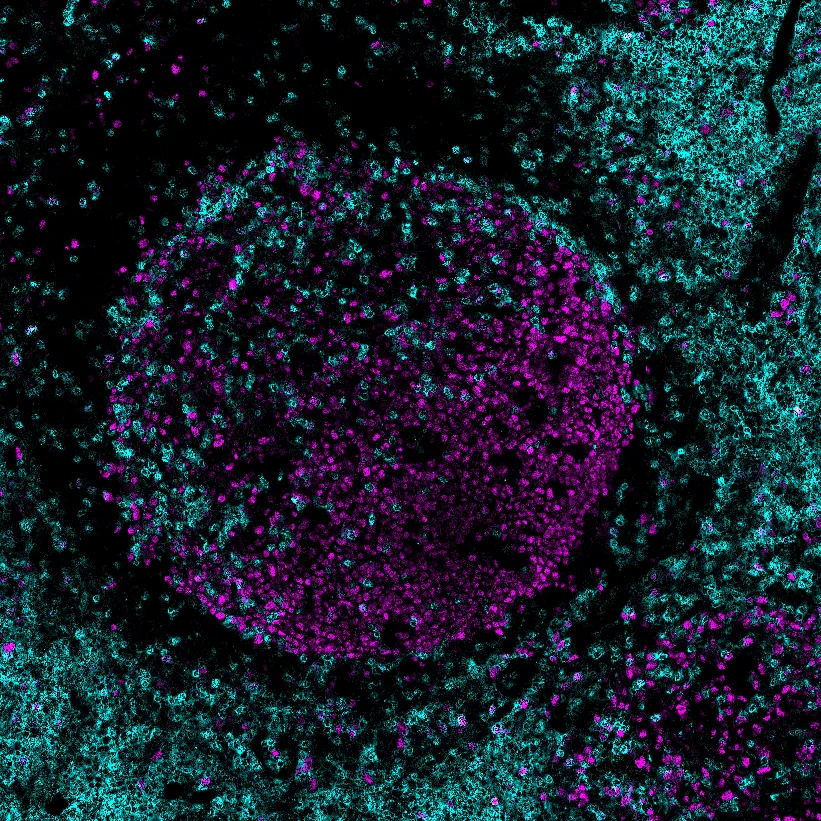
Sweet Germinal Centers
Germinal Centers (GC) are the central hubs where B cells fine-tune the affinity of their B cell receptor (BCR, membrane bound antibody). This affinity maturation is crucial for effective antibody responses as well as memory formation. It has long been known that activated GC stain positive with peanut agglutinin (PNA), while inactive follicles do not. Additionally, Solanum tuberosum agglutinin (STA) stains cellular subsets in the activated GC as well. Agglutinins bind specific sugar groups, such as sialic acids and other glycans. These sugar groups specifically change during active GC formation and glycans are known to signal in immune responses. This project focuses on the effect of sugar groups on adaptive immunity. The rationale for this project stems from one of our recent papers describing the follicular dendritic cell (FDC) transcriptome by single cell RNA sequencing. FDCs are stromal cells in the follicle with the capacity to regulate B and T cells. Our data revealed expression of several proteins involved in glycosylation.
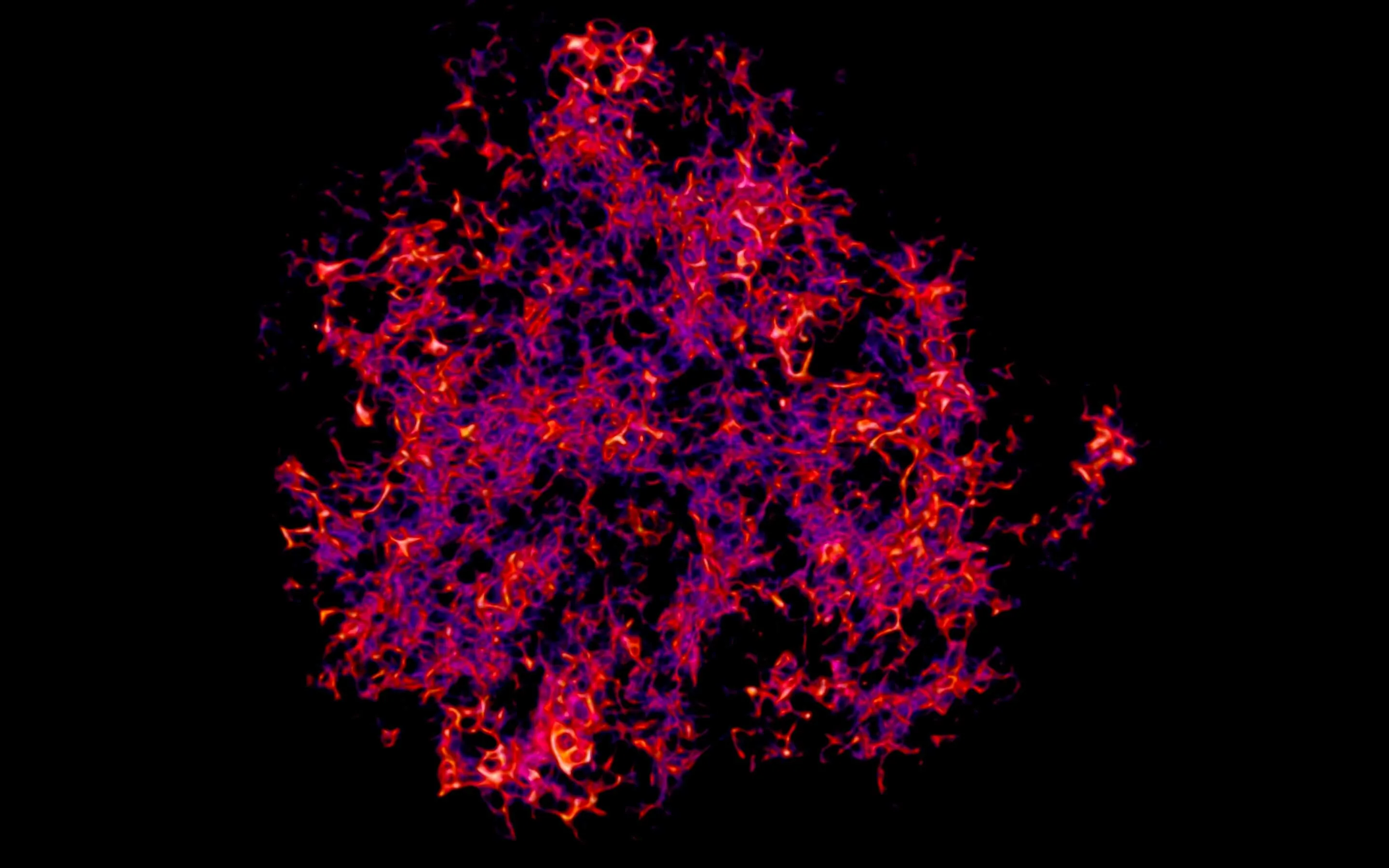
Targeting follicular dendritic cells as therapy:
Antigens are retained and presented to B cells by FDCs, but for therapeutic purposes it can be useful to displace this antigen or modify its fate. An important question is if self-antigen is retained and if it contributes to undesired immune responses. In this project we investigate the role of FDCs in auto-antibody production by developing sophisticated tools to intervene in antigen fate. Retention of antigen by FDCs is mediated mainly by Complement and its receptors. However, there are more players that can determine the fate of the antigen. Fc-receptors can bind immune complexes, and TLRs can be activated, which all can influence the fate of the immune complex. Interestingly, retention of proteins in endosomal compartments is partly regulated by sugar groups. Glycosylation could very well influence the shuttling of immune complexes within FDCs. Decoy complement receptors or antibodies targeting the complement-binding site can be used to dissociate antigen from the FDC, but a more elegant solution might be to modify glycosylation.
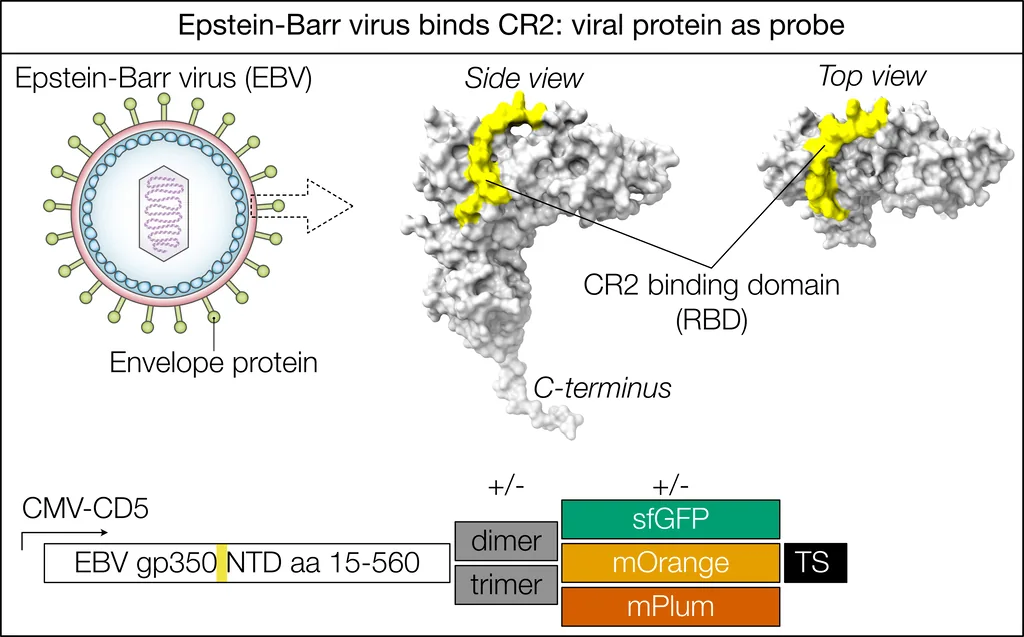
Block Epstein-Barr Virus with Complement (NWO ENW XS)
The exact cause of MS remains unclear, especially why and how the nervous system is attacked. The adaptive immune system is the most likely culprit, as immunosuppressive treatments help manage the disease. One theory suggests molecular mimicry: neurons might express a protein similar to one from the Epstein-Barr virus (EBV), causing the immune system to mistakenly attack neurons. However, this doesn't fully explain the periodic "flares" seen in relapsing-remitting MS, as you'd expect constant attacks on those neurons. An alternative hypothesis is that EBV latently infects specific neuron subsets. Herpes viruses like EBV periodically reactivate, which could explain the flare-ups and why only certain neurons are affected. For this to happen, neurons would need the EBV entry receptor (CR2), which is also found on B cells and some immune cells with neuronal traits (i.e., follicular dendritic cells).
"In short: EBV may trigger MS by infecting neurons, with periodic reactivation causing flare-ups."
Proofivy:
Our collaboration with Polygon to use popular blockchain technology in science is in its infancy and will continue to be developed in the coming years. The idea is to use blockchain to make scientific data verifiable and tamper-proof, which can help restore trust in science. By committing hashes of experimental setups, assumptions, hypotheses, and articles before publication, researchers can create a transparent and immutable record of their work. This can facilitate early collaboration and sharing of data while ensuring the integrity of scientific findings.
"Proofivy's mission is to increase trust in science, journalism, and humanity. Blockchain technology provides us with new tools for verification and storing of data. Proofivy makes it easy for researchers (and anyone else), to prove the creation and possession of data before they are ready to share it, and to store and share untamperable data."This concept was developed with a friend who worked at Polygon to help restore trust in science by making it verifiable. Together we are testing the concept and implementation. This can be used in the future to make early collaboration and sharing of data easier.
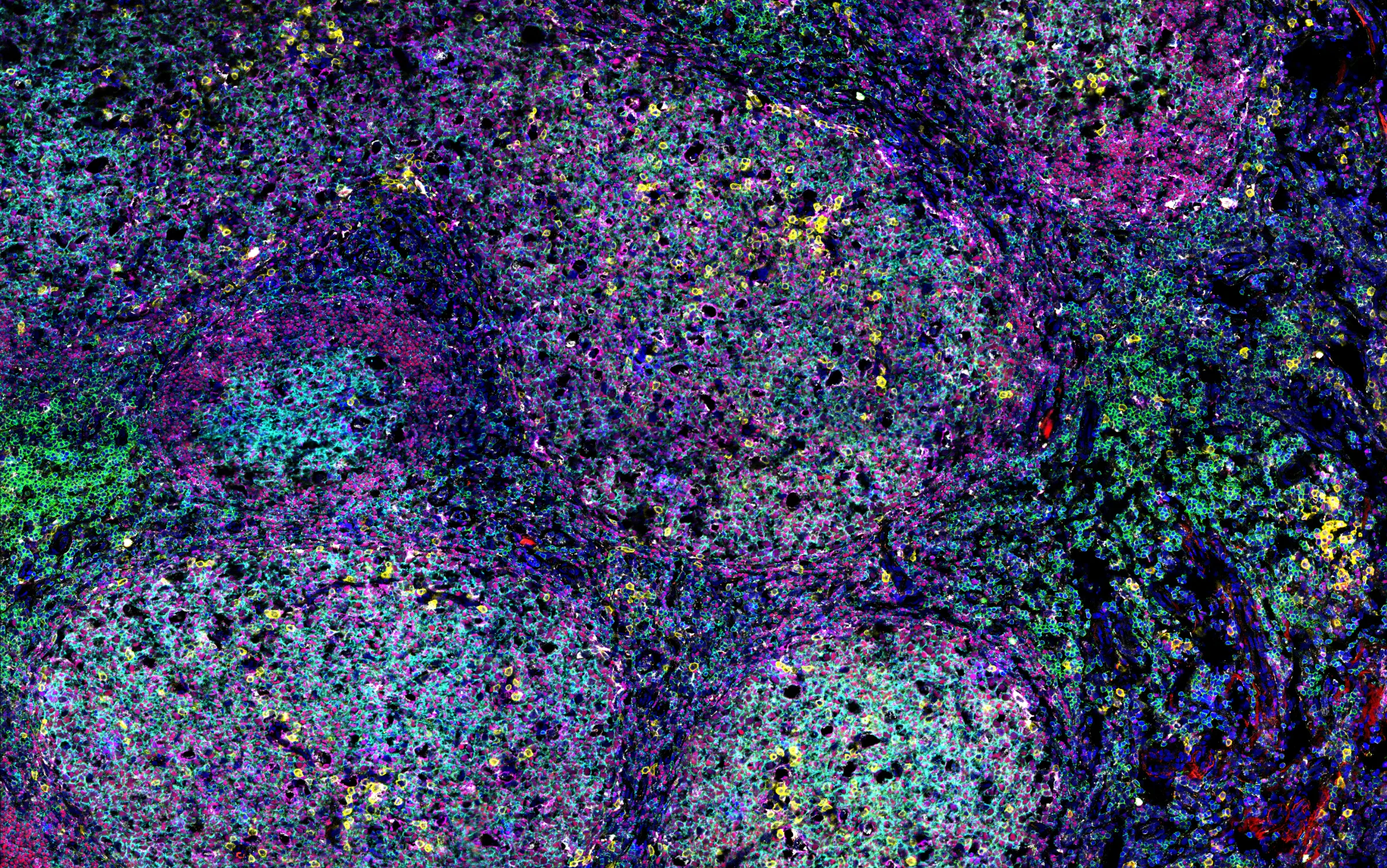
High-dimensional image analysis
To analyze complex tissues such as germinal centers, we use advanced microscopy techniques that generate large, multi-channel datasets. Manual analysis of these datasets is time-consuming and prone to bias. Therefore, we are developing computational pipelines that leverage machine learning and image analysis tools to segment and classify cell types, as well as quantify biologically relevant proteins and glycans within these tissues. This approach enables us to efficiently extract meaningful biological insights from high-dimensional imaging data. Use the slider to compare a raw microscopy image (left) with the corresponding segmented and classified image (right), generated by our analysis pipeline.
How projects are
Funded
Active funding
- Proefdiervrij Onderzoek grant
- GSK strategic partnership
- ZonMw VIDI grant
- Horizon Europe Doctoral Networks grant (Llimit consortium)
- ZonMw PharmaNL (PHARMIGA consortium)
Previous funding
- Ragon Innovation Award
- AMR postdoc fund
- NVVI work visit grant
- WBSO grant
- ZonMw VENI grant
- Polygon innovation fund
- NWO ENW XS grant
- Erasmus Scholarship to Melissa Ripamonti
- NWO Startersbeurs 2023